 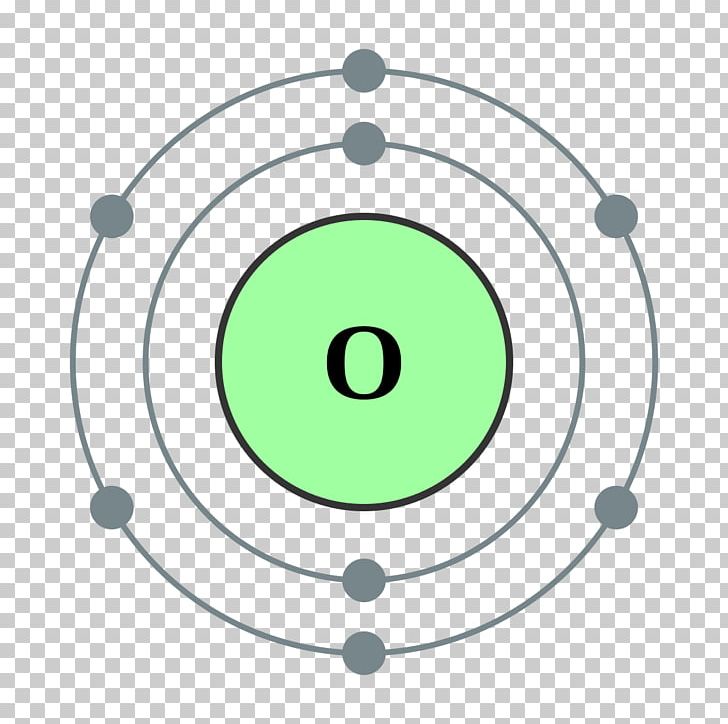
It is the basic unit of all light, carrying the energy E= hf. Photon: the smallest discrete amount of electromagnetic radiation. Ground state: is the lowest energy state of the atom.Įxcited-state: of an atom is a state where its potential energy is higher than the ground state.Įlectron: each electron has a negative charge (-1) with weight so small it’s normally negligible as compared to proton or neutron. 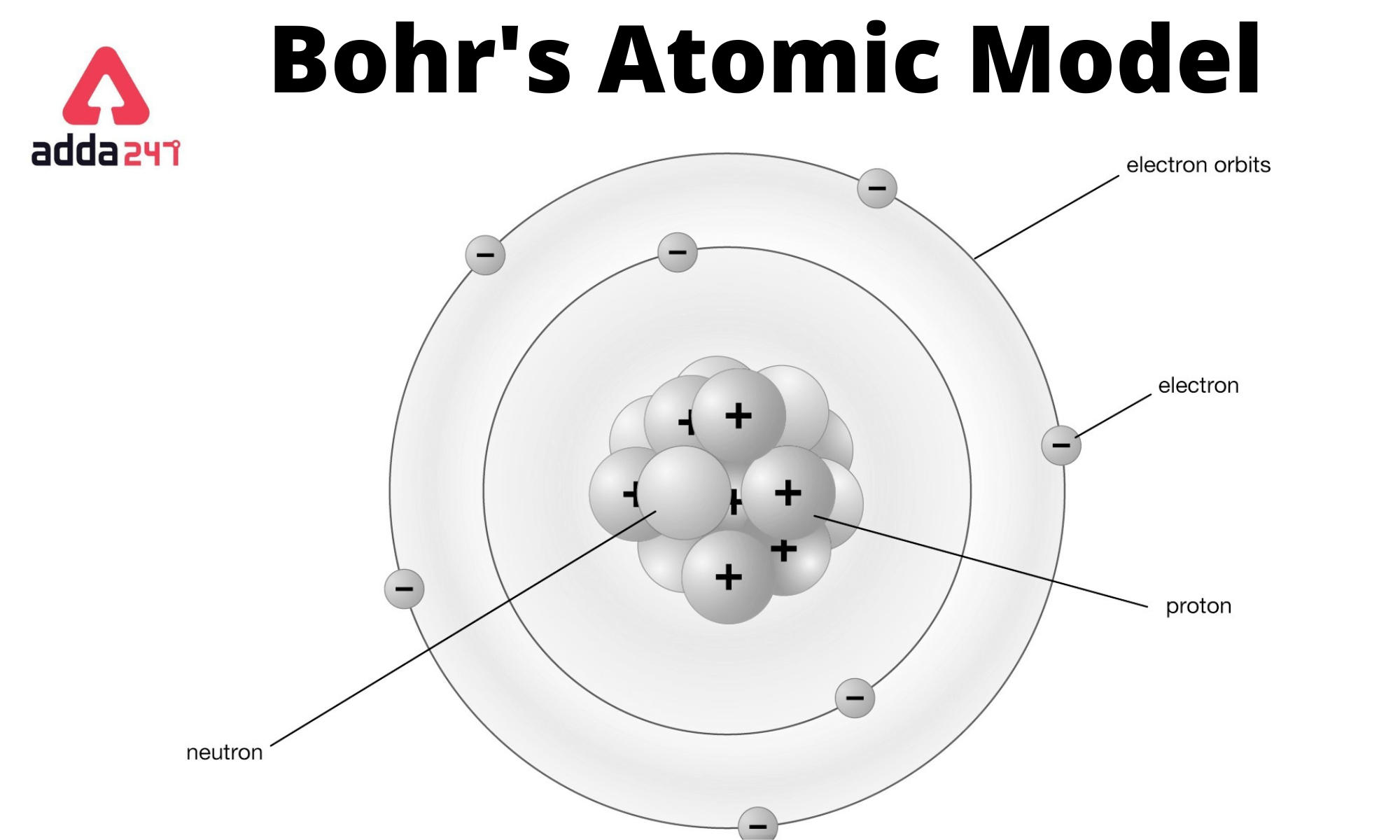
In 1913, Danish physicist Niels Henrik David Bohr improved Rutherford’s model by reasoning that if electrons were moving like planets, their orbits would be very unstable. Atoms consist of a nucleus surrounded by electrons. Bohr’s Model of the Atom Although Rutherford’s model was a breakthrough, it had some flaws, the main one being the way it described the motions of the electrons. The planetary model of the atom: or Bohr model of the atom, where the electrons are described to encircle the nucleus of the atom in specific allowable paths called the orbit.Ītom: The smallest possible amount of matter that still retains its identity as a chemical element. The electron is not allowed to occupy any of the spaces in between the orbits.Ītomic emission spectrum: is the pattern of lines corresponds to a different electron transition from a higher energy state to a lower energy state.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
